What is vaginal atrophy? This is a subject that doesn’t get talked about enough but that many women experience – the vaginal atrophy symptoms that can happen naturally with aging and menopause. This post includes a lot of research and experience to help anyone struggling with these challenges.
Let’s look at some of the current treatment options and cover one of my favorites (and often overlooked) – the use of the natural hormone DHEA – for effectively treating a more comprehensive range of vaginal atrophy symptoms.
We’ll explore the scientific research and clinical findings relating to the preferred vaginal atrophy treatment method, non-prescription topical DHEA cream.
Table of Contents
- Special NLF “Group Buy”: Julva FREE Trial!
- What Is Vaginal Atrophy?
- Current Treatment Options are Ineffective
- Lubricants & Vaginal Moisturizing Creams
- Kegel & Pelvic Floor Strengthening Exercises
- Surgical Options for Vulvar Rejuvenation
- DHEA – The Perfect Natural Solution
- Locally Applied DHEA to Treat Symptoms
- Dr. Anna’s Clinical Experience Using DHEA
- The Birth of Julva & Natural Living Family Special
What Is Vaginal Atrophy?
With aging, menopause, and hormonal changes some 75% of post-menopausal women suffer from vaginal atrophy (1) which can include,
- vulvar-vaginal thinning and irritation
- painful intercourse due to excessive vaginal dryness
- increased vaginal and bladder infections (due to decreased normal flora, loss of lactobacilli, and increased pH)
- leaking urine
- increased skin disorders affecting the urogenital tract (vulvodynia, vestibulitis, hypertrophic dystrophy, dermatitis)
- decreased desire, arousal, and orgasm
- pelvic prolapse symptoms such as pressure and trouble eliminating
Younger women can experience these symptoms as well.
Before menopause, a woman’s ovaries produce many of her sex hormones including estrogen. This female hormone maintains the health and integrity of the vaginal lining. During peri-post menopause, women experience declining hormone levels including estrogen, progesterone, testosterone, and dehydroepiandrosterone (DHEA).
Women’s ovaries gradually decrease producing these hormones causing vaginal lining shrinkage and drying due to decreased healthy secretions. Muscle loss also occurs. The lining becomes less elastic and thin, causing irritation and discomfort. The ovaries predominantly stop producing estrogen after menopause.
These and the other symptoms women experience from these decreasing hormones are life-affecting.
Stress urinary incontinence (SUI), the involuntary leakage of urine, has been estimated to affect over 50% of women between the ages of 20 to 80 years and was reported at 47% in a younger group consisting of women between 20 and 49 years of age. (2)
Women may suffer from decreased libido. The decline in androgens such as testosterone and DHEA are particularly associated with diminishing libido. (3, 4)
Increased pain during intercourse, increased post-coital infections and the decreased libido associated with vaginal atrophy have also been shown to increase the level of overall sexual distress in women. Sexual distress has been associated with a higher incidence of depression and relationship conflicts (5, 6) with more than 40% of women in the United States reporting sexual problems. (7)
Yet only 20% to 25% of symptomatic women having vaginal atrophy seek medical treatment. (8)
Lack of reporting symptoms to a doctor can be due to embarrassment, and/or a lack of awareness of vaginal health or treatment options. Women cannot readily examine their vulva or vagina, and can’t see the tissue changes that are otherwise easily visible to a physician. A physician can see extreme tissue changes in terms of color (pale or red), dryness, thinning, and irritation (red or raw patches).
Women may also mistake their symptoms for common irritations, allergies, or infections. More often the changes are so gradual, that the symptoms go unnoticed until there is pain, discharge, or incontinence.
The current life expectancy for American women is greater than 80 years old (as benchmarked in 2010 census data) and continues to increase. Given the average age of menopause, the onset is 50.5 years, it is estimated that women may expect to live almost 40% of their entire lives after menopause in this discomfort and decreased quality of life. (9)
Unlike hot flashes which usually end even without treatment, vaginal atrophy symptoms usually increase in severity over time. (10)
Decreasing hormones are also an important clinical health issue beyond vaginal health. Hot flashes, bone loss, fat accumulation, loss of muscle mass and strength (11), memory loss, cardiovascular disease, and type 2 Diabetes are additional concerns. (12)
Special 7-Day Sample Just for Natural Living Family Readers! With our Natural Living Family “group buy”, try a 7-day sampler of Julva, natural cream for delicate feminine tissue. Grab your FREE sample here.
Current Treatment Options are Ineffective
Traditional treatment options for vulvar-vaginal atrophy are only partially effective in addressing the many symptoms. Additionally, there are known safety risks.
Treatment for vulvar-vaginal irritation and pelvic support issues has been traditionally limited to lubricating creams and OTC options, herbal therapies, estrogen therapies, and other prescriptions, Kegel/pelvic-strengthening exercise, and surgery.
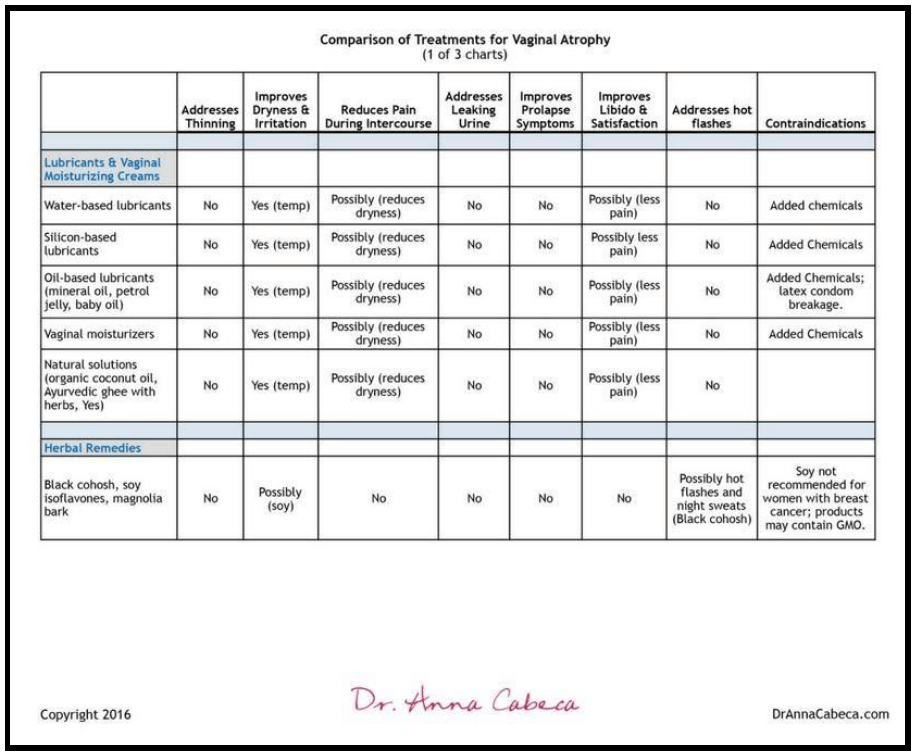
Lubricants & Vaginal Moisturizing Creams
While helpful for lubrication these products are primarily cosmetic and do not treat underlying concerns. They may help with irritation and painful intercourse due to reducing dryness.
- Water-based lubricants include: K-YJelly, FemGlide, Summer’s Eve and others are non-staining. Silicone-based lubricants include Pink, Pure Pleasure, and others.
- Oil-based lubricants include using mineral oil, petroleum jelly, or baby oil. These are not recommended as they can actually cause irritation and have also been associated with high rates of latex condom breakage.
- Vaginal moisturizers include Replens, Moist Again, Fresh Start, and K-Y Liquibeads.
Natural solutions such as organic coconut oil-based homemade lubricants, Ayurveda ghee (combined with herbs), and an OTC organic lubricant are also available and do not contain the additional chemicals that can be seen in other commercial lubricants and vaginal moisturizing solutions.
Herbal therapies have been used and a number of OTC herbal remedies are available containing black cohosh, soy isoflavones, magnolia bark, and other ingredients.
The American College of Obstetricians and Gynecologists (ACOG) reports a concern that many of the early studies of black cohosh were poorly designed and did not evaluate its safety and effectiveness beyond 6 months of use. (13)
A soy-rich diet has been shown to be helpful in some cases, but more research is needed. (14)
Soy products – like Estroven and Remifemin – may not be appropriate for women having breast cancer. Also, some doctors have advised women to only use non-GMO fermented soy foods such as miso or tempeh in moderation.
This chart summarizes the above treatment options:
Estrogen Therapies for Vaginal Atrophy
One common treatment for vaginal atrophy is to replace the diminished estrogen levels through a variety of potential chemicals, creams, or other means.
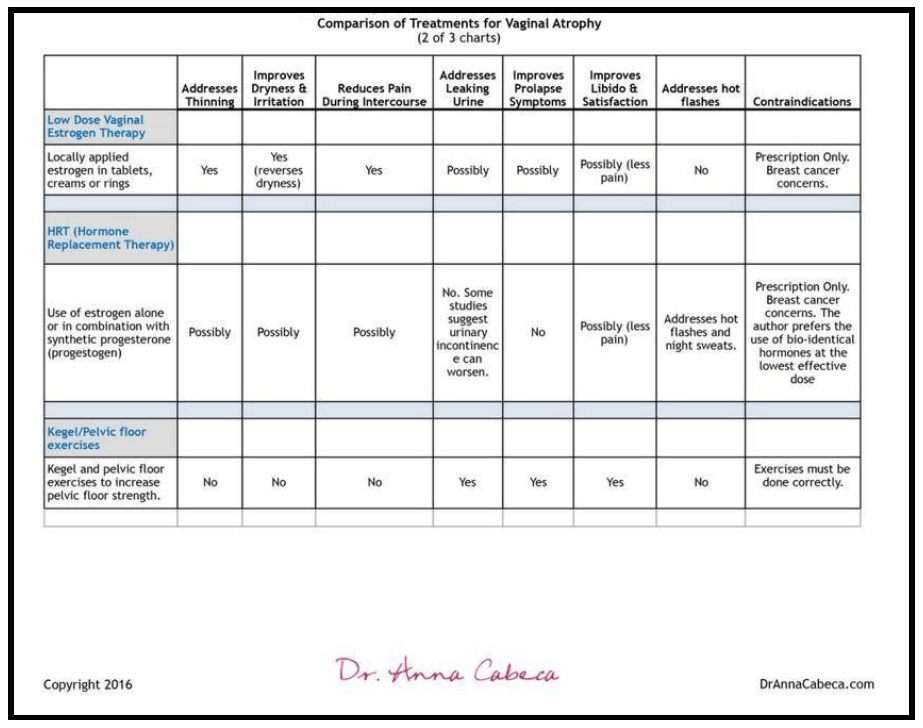
Low-Dose Vaginal Estrogen Therapy
Predominant medical therapy of vaginal dryness and pain has been with vaginal estrogen.
This therapy can be prescribed as vaginal tablets, creams, or rings. These are delivered locally within the vagina and therefore minimize estrogen increases in blood levels along with related possible side effects. They help improve the thickness and elasticity of the vaginal lining but do not affect the deeper tissue or supporting muscles, nor does research show that they address incontinence issues.
Examples include prescription medications, Vagifem, Premarin, Estring, and Estrace. Due to the estrogen content, vaginal estrogen therapy is not recommended for some women. Women with breast cancer or women with a history of stroke or heart attack, blood clots, or liver disease should not opt for estrogen replacement therapies. (15)
Hormone Replacement Therapy (HRT) & Vaginal Health
Estrogen products, alone or in combination, that raise levels of the hormone throughout the body (systemically), not just in the vagina, are referred to as “hormone replacement therapy (HRT)”. These products address vaginal atrophy and related pain during sex in postmenopausal women as well as addressing other common symptoms of menopause such as hot flashes and night sweats.
Despite their benefits on vasomotor symptoms, 40% of women receiving systemic estrogen therapy have persistent vaginal symptoms. (16) Additionally, some studies conclude that HRT has been associated with worsening urinary incontinence. (17)
- HRT, typically not bio-identical including estrogen and progestins (differs from bio-identical progesterone) is not known to address libido although pain during sexual intercourse may be diminished.
- HRT therapy has been shown to help prevent osteoporosis. (18)
- Hormone replacement therapy prescriptions may contain estrogen alone or may include progestogen (synthetic progesterone) along with estrogen. There are known risks relating to increased risk of blood clots, breast cancer, heart attack, and stroke with oral administration.
My friend, Dr. Anna, has a strong preference from clinical and scientific experience and research that hormone replacement should only be bio-identical and in the lowest effective dose initially to achieve optimal results.
SPECIAL PRICE JUST FOR NATURAL LIVING FAMILY READERS!
This Natural Living Family group buy for Julva, a cream that soothes, repairs, and rejuvenates the feminine you. Get your trial pack here (just pay shipping)!
Kegel & Pelvic Floor Strengthening Exercises
Kegel exercises strengthen the pubococcygeus muscle and can improve symptoms of incontinence, as well as arousal and orgasm. They can also prevent or improve symptoms of pelvic organ prolapse, in which the uterus or bladder bulges into the vagina due predominantly to muscle weakness.
- Kegel exercises involve contracting and relaxing the muscles of your pelvic floor, which holds your uterus and bladder above your vagina.
- Women can also use jade balls/Lelo balls/kegel balls.
- It is also important to avoid exercises that cause increased pressure on the pelvic floor.
Here is a summary of the estrogen treatment options, as well as the pelvic floor health (Kegels) options:
Surgical Options for Vulvar Rejuvenation
Surgical options have traditionally included specific surgeries focused on vulvar rejuvenation as well as those to support pelvic support and incontinence issues. These are currently viewed as two different categories of surgical intervention, one for functional health reasons (incontinence and prolapse) and one viewed primarily as cosmetic.
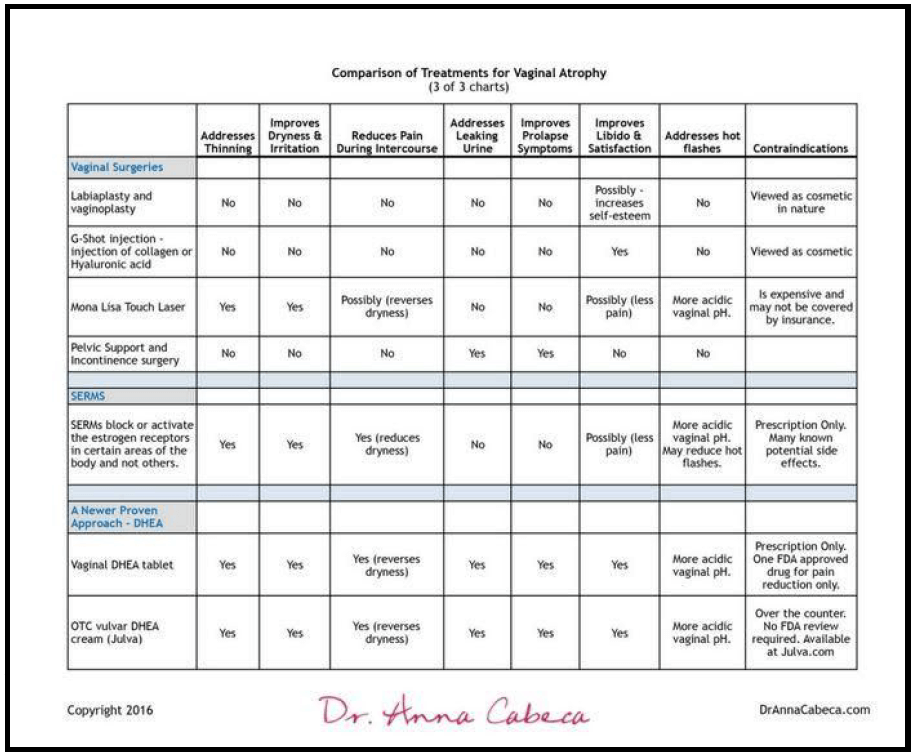
1. Labiaplasty & Vaginoplasty Viewed as Primarily Cosmetic.
Many women view these procedures as important for their self-esteem and sexual satisfaction. However, most mainstream health organizations still consider them as cosmetic.
American Society of Aesthetic Plastic Surgery (ASAPS) released statistics in 2014 that revealed a 49% increase (from 5,070 surgeries to 7.535) in labiaplasty and other genital cosmetic procedures. (19) A detailed review of these procedures can be found at https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3312147/.
The American Congress of Obstetricians and Gynecologists have stated in 2007, and reaffirmed in 2014 (20) that,
“So-called “vaginal rejuvenation,” “designer vaginoplasty,” “revirgination,” and “G-spot amplification” are vaginal surgical procedures being offered by some practitioners. These procedures are not medically indicated, and the safety and effectiveness of these procedures have not been documented. Clinicians who receive requests from patients for such procedures should discuss with the patient the reason for her request and perform an evaluation for any physical signs or symptoms that may indicate the need for surgical intervention. Women should be informed about the lack of data supporting the efficacy of these procedures and their potential complications, including infection, altered sensation, dyspareunia, adhesions, and scarring.”
G-shot injection
This is the injection of collagen or Hyaluronic acid injected into the G spot. 87% of recipients in one pilot study reported increased sexual arousal and gratification. (21)
2. Pelvic Support & Incontinence Surgery: Viewed as Addressing Functional and Urinary Incontinence Issues.
MonaLisa Touch Laser Therapy
This vaginal laser treatment was approved by the U.S. Food and Drug Administration (FDA) for aesthetic use in 2014, including approval for gynecologic use. (22)
The laser technology results in tissue regeneration and addresses several symptoms relating to vaginal atrophy. In particular, the vaginal mucosa tissue becomes more nourished and hydrated; the epithelium becomes thicker and regains some elasticity. It also reestablishes a more acidic vaginal pH. It is not FDA approved for treating urinary incontinence. The procedure is relatively new, and is not normally covered by insurance, and can be expensive.
Pelvic Prolapse & Incontinence Surgeries
Research prior to 2014 has shown that about 3% of U.S. women will have symptoms of prolapse in a given year. According to the authors of one study published in 2014, in the Journal of the American Medical Association, about 300,000 U.S. women undergo surgery for prolapse every year. (23)
The two most common surgeries are uterosacral ligament suspension and sacrospinous ligament fixation. This study, partly funded by the National Institute of Health Office of Research on Women’s Health, found positive outcomes for both treatments relating to prolapse symptoms and a small risk of side effects.
While these procedures may address pelvic prolapse and incontinence issues they do not address many of the other major symptoms of vaginal atrophy including dryness and itching, pain with intercourse, or libido/sexual satisfaction.
Non-Traditional Treatment Options include SERMs & DHEA
Less known hormone therapy treatment options include selective estrogen receptor modulators (SERMs) and the use of DHEA.
SERMs
Women suffering from vaginal atrophy may utilize SERMs, rather than traditional estrogen therapies, for relief. SERMs block or activate the estrogen receptors in certain areas of the body and not others. This can make them safer than estrogen therapy alone especially in a woman having a history or family history of breast cancer. SERMs can also relieve other menopausal symptoms such as hot flashes and even bone density.
SERMS with positive vulvar-vaginal effects include lasofoxifene and ospemifene. These SERMs improve vaginal atrophy and reduce vaginal pH and more, but do have potentially serious side effects. (24) More research is ongoing but this is an exciting new area of addressing vaginal atrophy.
Special 7-Day Sample Just for Natural Living Family Readers! With our Natural Living Family “group buy”, try a 7-day sampler of Julva, natural cream for delicate feminine tissue. Grab your FREE sample here.
DHEA – The Perfect Natural Solution
While most research and available treatments have been focused on the effects of diminishing estrogen and testosterone, there is another key hormone that has been found to play an important role in addressing vaginal atrophy symptoms, and that is dehydroepiandrosterone (DHEA).
DHEA is an androgen, like testosterone. Androgens are important to the integrity of skin, muscle, and bone (in both males and females) and have a role in maintaining libido. They also improve energy level and mental alertness, provide cardiovascular protection by lowering cholesterol and enhance bone building (by increasing calcium retention).
DHEA originates from the adrenal gland. It is an inactive precursor that leads to the production of active sex hormones like androgens or estrogens in specific cells and tissues. As estrogen levels naturally decrease it is DHEA that continues to be a remaining source of estrogens and androgens in the woman’s body.
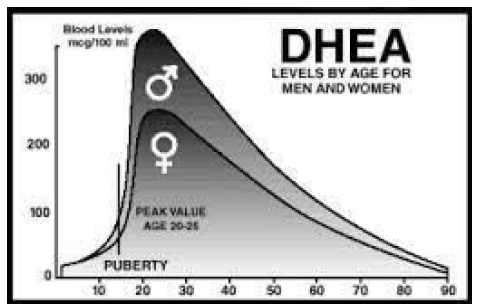
DHEA is produced by the body naturally and “extends” protective benefits to women as their estrogen levels decrease, but only for a time. There is a progressive decrease in serum DHEA which starts at the age of 30 years with an average 60% loss observed by menopause. (25)
DHEA, however, can be introduced and utilized by the body. DHEA can be introduced orally, vaginally or through topical application around the vulva.
Natural decline of DHEA
Oral DHEA
While used for a variety of other health benefits, Oral DHEA has not been shown to address vaginal atrophy symptoms. (26)
Locally Applied DHEA
Topical DHEA has been found to have favorable effects on skin health and appearance (27) due to the production of collagen.
If DHEA is delivered directly to the vagina, the tissues transform DHEA to estrogen, estradiol. (28) This natural production of estradiol occurs without a significant release of estrogens systemically in the blood.
Dr. Anna and other physicians have been able to write prescriptions for customized vaginal and topical DHEA and other hormones for decades with many positive results.
In November 2016 the U.S. Food and Drug Administration (FDA) approved the first product containing the active ingredient Prasterone, also known as DHEA. (29) The product, Intrarosa®, was approved to treat women experiencing moderate to severe pain during sexual intercourse, a chief symptom of vulvar atrophy.
Here is a summary of surgical options, SERMS, and DHEA.
Locally Applied DHEA to Treat Symptoms
There is a good deal of research on the benefits and effectiveness (as well as safety) of DHEA in treating vulvar-vaginal symptoms.
The research surrounding vaginally applied DHEA has shown it to:
- Reduce vaginal dryness and irritation
- Strengthen vaginal musculature
- Increase bone mineral density
- Decrease pain during intercourse
- Increase arousal and libido, as well as sexual satisfaction
For women who have had breast cancer: There has also been some exciting research done (not yet published) that has shown vaginally applied DHEA to effectively treat vaginal dryness, pain, and other vaginal atrophy symptoms – including libido. This 2014 clinical trial has not yet been published but there is more information included on Dr. Anna’s website should you be interested in learning more.
1. Reduce Vaginal Dryness & Irritation
In a study reported in the Journal of The North American Menopause Society (30) in 2016, daily intravaginal administration of DHEA caused highly statistically significant improvements in four measurements of vaginal atrophy.
| 482 Participants using DHEA (0.50% daily Prasterone for 12 weeks) | |
| Vaginal dryness
improvements |
1.44 severity score units compared to baseline, or 0.27 units over placebo |
| Gynecological evaluations | 86% to 121% improvements (4 measures) over the placebo |
| Vaginal pH acidity | Decreased by 0.66 pH units over placebo |
| Pain during sexual activity | Decreased by 1.42 severity score units from baseline or 0.36 units over placebo |
2. Strengthen Vaginal Musculature
In another 2009 study, DHEA was applied locally within the vagina resulting in a significant improvement to all three layers of the vagina (epithelium, lamina propria, and muscularis) versus simply affecting the superficial epithelial cells. (31)
Dr. Anna has numerous case studies demonstrating improvements in vaginal musculature as well as urinary incontinence issues through the use of locally applied DHEA.
3. Increase Bone Mineral Density
In clinical studies, locally applied DHEA has been found to increase bone mineral density (32) and to result in an increase in serum osteocalcin, a marker of bone formation. (33)
4. Decrease Pain During Intercourse (Dyspareunia)
In a clinical trial in 2015 using intravaginal Prasterone (6.5 mg daily for 12 weeks) there was a statistically significant beneficial effect on moderate to severe dyspareunia. (34) It’s also been noted that vaginal dryness and dyspareunia presented together in 70-80% of women.
5. Increase Arousal & Libido (as well as sexual satisfaction)
In the 2009 study, 1.0% (13 mg) DHEA applied locally within the vagina resulted in a marked improvement to four aspects of sexual dysfunction: desire, arousal, pleasure, and orgasm. (35)
| 1.0% vaginal DHEA over a 12 week period | |
| Improvements in desire | 23% versus placebo |
| Improvements in arousal and lubrication | 139% versus placebo |
| Improvements to orgasm | 75% versus placebo |
| Improvements in dryness during intercourse | 57% versus placebo |
In a 2015 study the long-term effect on the sexual function of 154 postmenopausal women reporting some form of vaginal atrophy was evaluated based on a 52-week treatment with daily intravaginal 0.50% (6.5 mg) DHEA. pubococcygeus
Results showed increases as follows: desire, arousal, lubrication, orgasm, satisfaction and pain were improved by 28%, 49%, 115%, 51%, 41% and 108%, respectively. (36)
Special 7-Day Sample Just for Natural Living Family Readers! With our Natural Living Family “group buy”, try a 7-day sampler of Julva, natural cream for delicate feminine tissue. Grab your FREE sample here.
Dr. Anna’s Clinical Experience Using DHEA
As an Emory-trained physician, Dr. Anna started using androgen therapy in her private practice in 1999. She used bioidentical DHEA and Testosterone vaginally or applied it topically to the vulvar area. Patients came to her complaining of vaginal atrophy symptoms including vaginal dryness, irritation, pain during intercourse, and urinary leakage. Many suffered from libido issues.
She routinely achieved positive results with her patients for improvements to sexual health and vaginal dryness, and a reduction in irritation and pain during intercourse. There were regular improvements in libido and sexual satisfaction. Some patients also reported decreased incontinence symptoms. Many patients seeing her for possible surgery due to pelvic prolapse and stress incontinence issues were able to avoid surgery altogether once she prescribed vaginal DHEA and testosterone to them.
For these patients, a physical examination verified their reported improvements; there were notable tissue improvements in the lining and within the vaginal ruggations (the normal folds and elasticity) as well as contractility (the ability to contract the muscles of the pelvic floor). This meant more strength, more support to the urethra and bladder too.
The Birth of Julva & Natural Living Family Special
Dr. Anna wanted to be able to provide a non-prescription based topical solution that provided the least systemic effect. This would provide women all over the world with a less embarrassing option to address a significant quality of life and overall health issue associated with aging.
Three years of research along with her extensive clinical results led me to combine DHEA in cream form with other quality natural ingredients having been shown to be beneficial to the skin and its underlying tissues. Those ingredients include Alpine Rose Stem Cells, Emu oil, Vitamin E Tocopherol, Coconut oil, and Shea Butter.
Alpine Rose Stem Cells are harvested from Swiss alpine plants and contain unique compounds that help the plant survive extremely challenging environments. The stem cells have been shown to increase skin cell replenishment, protect against age-related oxidative stress, have antiviral effects and are loaded with polyphenolic antioxidant compounds.
Vitamin E Tocopherol provides anti-inflammatory support; Coconut oil provides a safe and natural lubricant; Emu oil promotes deeper tissue absorption and regenerates skin cells while improving thinning skin.
Her research concluded that a small 5-10 mg daily dosage of DHEA topically applied achieved excellent symptom improvements, after which she recommends a reduced maintenance dosage protocol.
Dr. Anna perfected this cream with love and prayers that it helps women around the world feel good about their sexual health especially as we deal with the normal changes of age. We’ve shared it with our Natural Living Family in the past and the testimonials have been empowering.
We love that there was such a focus on making it really safe, and along with addressing all of the issues above…it is designed to effectively improve the divinely designed function of our beautiful, feminine, pelvic floor. You don’t have to suffer from these symptoms any longer and you don’t have to resort to unsafe treatments to find relief. Please try my friend Dr. Anna’s safe and effective cream and see how it can help.
References:
1. Fernand Labrie, MD, PhD, David Archer, MD, Ce´line Bouchard, MD, et al. Intravaginal dehydroepiandrosterone (Prasterone), a physiological and highly efficient treatment of vaginal atrophy. The Journal of the North American Menopause Society. 2009;16(5):907-922.
2. Patrick J. Culligan, MD, and Michael Heit, MD, University of Louisville Health Sciences Center, Louisville, Kentucky. Urinary incontinence in women: evaluation and management. Am Fam Physician. 2000; Dec 1;62(11):2433-2444.
3. S. Leiblum, G. Bachmann, E. Kemmann, D. Colburn, L. Schwartzman. Vaginal atrophy in the postmenopausal woman. The importance of sexual activity and hormones. JAMA. 1983;249(16):2195-2198. hormones.
4. N.E. Avis, S. Brockwell, J.F. Randolph Jr, et al. Longitudinal changes in sexual functioning as women transition through menopause: results from the Study of Women’s Health Across the Nation. Menopause. 2009;16(3):442-452.
5. L. Dennerstein, J.R. Guthrie, R.D. Hayes, L.R. DeRogatis, P. Lehert. Sexual function, dysfunction, and sexual distress in a prospective, population-based sample of mid-aged, Australian-born women. J Sex Med. 2008 Oct;5(10):2291-9. Epub 2008 Jul 14.
6. L.R. Knoepp, S.H. Shippey, C.C. Chen, G.W. Cundiff, L.R. DeRogatis, V.L. Handa. Sexual complaints, pelvic floor symptoms, and sexual distress in women over forty. J Sex Med. 2010 Nov;7(11):3675-82.
7. Jan L. Shifren, MD; Brigitta U. Monz, MD; Patricia A. Russo, PhD; Anthony Segreti, PhD; Catherine B. Johannes, PhD. Sexual problems and distress in United States women: prevalence and correlates. Obstetrics & Gynecology. 2008 November;112(5):970-978.
8. Fernand Labrie, MD, PhD, David Archer, MD, Ce´line Bouchard, MD, et al. Intravaginal dehydroepiandrosterone (Prasterone), a physiological and highly efficient treatment of vaginal atrophy. The Journal of the North American Menopause Society. 2009;16(5):907-922.
9. Sharon J. Parish, Rossella E. Nappi, Michael L. Krychman, et al. Impact of vulvovaginal health on postmenopausal women: a review of surveys on symptoms of vulvovaginal atrophy. Int J Womens Health. 2013;5:437-447.
10. J. Calleja-Agius, M.P. Brincat. Urogenital atrophy, Climacteric. 12 (4) (2009) 279–285.
11. Labrie F, Diamond P, Cusan L, Gomez JL, Be´langer A, Candas B. Effect of 12-month dehydroepiandrosterone replacement therapy on bone, vagina, and endometrium in postmenopausal women. J Clin Endocrinol Metab. 1997;82:3498-3505.
12. DT Villareal, JO Holloszy. Effect of DHEA on abdominal fat and insulin action in elderly women and men: a randomized controlled trial. JAMA 2004;292:2243-2248.
13. Available at: http://umm.edu/health/medical/altmed/herb/black-cohosh. Accessed January 2, 2017.
14. L.M. Chiechj, G. Putignano, V. Guerra, M.P Schiavelli, A.M. Cisternino, C. Carriero. The effect of a soy-rich diet on the vaginal epithelium in postmenopause: a randomized double-blind trial. Maturitas. 2003. Aug 20;45(4):241-6.
15. Rippy L, Marsden J. Is HRT justified for symptom management in women at higher risk of developing breast cancer? Climacteric. 2006;9:404–15.
16. S.L. Johnston, S.A. Farrell, C. Bouchard, et al. The detection and management of vaginal atrophy. J Obstet Gynaecol Can 2004;26:503-515.
17. Jay H. Lee, MD, Suzanne Gomez, MD, Terry Ann Jankowski, MLS. Hormone therapy for postmenopausal women with urinary incontinence. Am Fam Physician. 2011 Jul 1;84(1).
18. LP Shulman. Transdermal hormone therapy and bone health. Clin Interv Aging. 2008;3:51–4. Copyright 2016 (updated 8/17)Dr. Anna Cabeca DrAnnaCabeca.Com
19. Available at: http://asj.oxfordjournals.org/genitalrejuvenation. Accessed January 2, 2017.
20. Available at: http://www.acog.org/Resources-And-Publications/Committee-Opinions/Committee-on-Gynecologic-Practice/Vaginal-Rejuvenation-and-Cosmetic-Vaginal-Procedures. Accessed January 2, 2017.
21. Available at: www.thegshot.com. Accessed January 2, 2017.
22. Available at: https://www.acog.org/-/media/434ADADB30E846B09D359F3F1432510E.pdf Fractional laser treatment of vulvovaginal atrophy and U.S. Food and Drug Administration clearance position statement The American College of Obstetricians and Gynecologists and The American Congress of Obstetricians and Gynecologists. May 2016. Accessed January 2, 2017. Copyright 2016 (updated 8/17)Dr. Anna CabecaDrAnnaCabeca.Com
23. Barber, M. D., L. Brubaker, K.L. Burgio, H.E. Richter, I. Nygaard, S.F. Meikle; Eunice Kennedy Shriver National Institute of Child Health and Human Development Pelvic Floor Disorders Network (2014). Comparison of two transvaginal surgical approaches and perioperative behavioral therapy for apical vaginal prolapse: the OPTIMAL randomized trial. JAMA, 311(10), 1023–1034.
24-25. Labrie F, Be´langer A, Cusan L, Gomez JL, Candas B. Marked decline in serum concentrations of adrenal C19 sex steroid precursors and conjugated androgen metabolites during aging. J Clin Endocrinol Metab 1997;82:2396-2402.
26. R.M. Goel, A.R. Cappola. Dehydroepiandrosterone sulfate and postmenopausal women. Curr Opin Endocrinol Diabetes Obes. 2011 Jun;18(3):171-6.
27. Available at: http://www.lifeextension.com/magazine/2010/12/new-research-substantiates-the-anti-aging-properties-of-dhea/page-02. Accessed January 2, 2017.
28. Labrie F, Bélanger A, Bélanger P, et al. Androgen glucuronides, instead of testosterone, as the new markers of androgenic activity in women. J Steroid Biochem Mol Biol. 2006;99:182–8.
29. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm529641.htm. Accessed January 2, 2017.
30. F. Labrie, D.F. Archer, W. Koltun, et al. Efficacy of intravaginal dehydroepiandrosterone (DHEA) on moderate to severe dyspareunia and vaginal dryness, symptoms of vulvovaginal atrophy, and of the genitourinary syndrome of menopause. Menopause. 2016 Mar;23(3):243-256.
31. F. Labrie, D.F. Archer, C. Bouchard, et al. Effect of intravaginal dehydroepiandrosterone (Prasterone) on libido and sexual dysfunction in postmenopausal women. Menopause: The Journal of the North American Menopause Society . 2009 Sept-Oct;16(5):923-31.
32. F. Labrie, V. Luu-The, C. Labrie, et al. Endocrine and intracrine sources of androgens in women: inhibition of breast cancer and other roles of androgens and their precursor dehydroepiandrosterone. Endocr Rev. 2003 Apr;24(2):152-82.
33. F. Labrie, P. Diamond, L. Cusan, J.L. Gomez, A. Be´langer, B. Candas. Effect of 12-month dehydroepiandrosterone replacement therapy on bone, vagina, and endometrium in postmenopausal women. J Clin Endocrinol Metab. 1997;82:3498-3505.
34. D.F. Archer, F. Labrie, C. Bouchard, et al. Treatment of pain at sexual activity (dyspareunia) with intravaginal dehydroepiandrosterone (Prasterone). Menopause: The Journal of The North American Menopause Society. 2015 Sep;22(9):950-63.
35. F. Labrie, D.F. Archer, C. Bouchard, et al. Effect of intravaginal dehydroepiandrosterone (Prasterone) on libido and sexual dysfunction in postmenopausal women. Menopause: The Journal of the North American Menopause Society. 2009 Sept-Oct;16(5):923-31.36 C. Bouchard, F. Labrie, L. Derogatis, VVA Prasterone Group, et al. Effect of intravaginal dehydroepiandrosterone (DHEA) on the sexual function in postmenopausal women: ERC-230 open-label study. Horm Mol Biol Clin Investig. 2016 Mar;25(3):181-90.
36. Available at: https://mibellebiochemistry.com/media/471/download. Accessed January 2, 2017.